|
For example, air, sea water, crude oil, etc. That means that it can not be separated into its constituents by mechanical or physical means and only can be destroyed by chemical means.Ī mixture is a material containing two or more elements or compounds that are in close contact and are mixed in any proportion. 
In essence, an element is a substance consisting of one type of atom.Ī compound is a pure substance composed of two or more different atoms chemically bonded to one another. An element is a substance that can not be further reduced as to simpler substances by ordinary processes. If we are to understand this diversity, we must start with a way of organizing and describing matter.Īll matter is made of elements that are fundamental substances that cannot be broken down by chemical means. The diversity of the matter in the world and in the universe is astounding. To better understand this matter-how it affects you, how you affect it and how it can be manipulated to our benefit-we need get a basic understanding of the types and properties of matter. We are all completely surrounded by matter. Give some applications of non-metal alloys.Explain what metal alloys are and explain the significance of metal alloys in material science and material engineering.Explain how material and mechanical engineers use these terms regarding creating new composite materials.Explain how chemical engineers use these terms when solving problems related to water purification and distillation of crude oil.Explain the different properties of each group of materials.Give some examples of elements, mixtures, and compounds.Distinguish and describe the three types of matter: elements, compounds, mixtures.Understanding the properties of a given material, element, component, or composite is a critical part of the engineering design process.Īfter this activity, students should be able to: They take advantage of the varying strengths and abilities of different materials to make composites with significantly different physical or chemical properties. Materials scientists and mechanical engineers focus on understanding the nature and properties of varying materials so that they can iterate upon them and create more robust products. For example, bitumen (a homogeneous mixture) is a component of asphalt (a heterogeneous mixture).Copyright © A homogeneous mixture can be a component of a heterogeneous mixture.Some mixtures that appear homogeneous at first glance are heterogeneous upon closer inspection.Examples include orange juice with pulp and chicken noodle soup. Many familiar items are heterogeneous mixtures.Examples include vodka, vinegar, and dishwashing liquid. Many common chemicals are homogeneous mixtures.For example, you can make a homogeneous solution of sugar and water, but if there are crystals in the solution, it becomes a heterogeneous mixture. The exception would be solutions that contain another phase of matter. Chemical solutions are usually homogeneous mixtures.A good example is a mixture of oil and water. 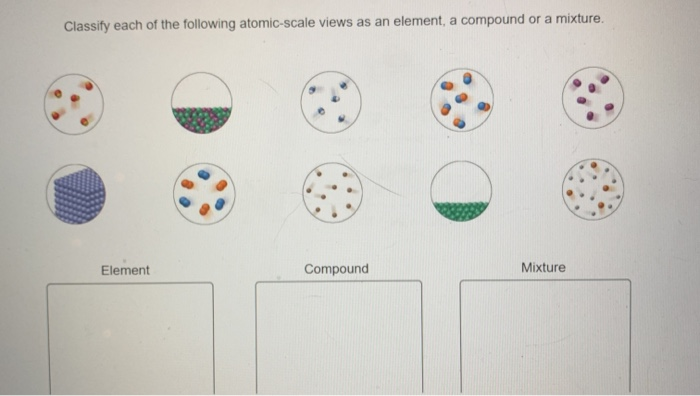
The liquid that is immiscible form heterogeneous mixtures.Examples include ice cubes in a drink, sand and water, and salt and oil. Mixtures in two or more phases are heterogeneous mixtures. 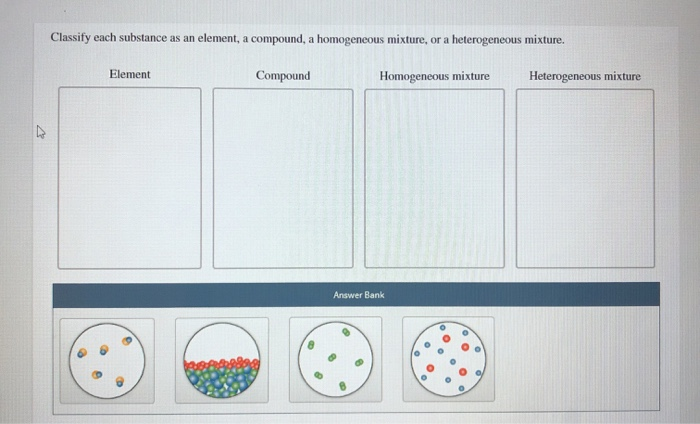
Examples include sand and sugar, salt and gravel, a basket of produce, and a toy box filled with toys. Mixing together two solids, without melting them together, typically results in a heterogeneous mixture.The two types of mixtures are distinguished by the size of the crystals that are present. In these cases, they are heterogeneous mixtures. Sometimes multiple phases exist in alloys. Examples include brass, bronze, steel, and sterling silver. Alloys are made when two or more metals are mixed together.See the clouds? That's evidence the composition is not uniform. However, the Earth's atmosphere as a whole is a heterogeneous mixture.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
